Exploring the Conformational Transitions of Biomolecular Systems Using a Simple Two-State Anisotropic Network Model
By Avisek Das, Mert Gur, Mary Hongying Cheng, Sunhwan Jo, Ivet Bahar, and Benoît Roux.
Published in PLoS Computational Biology 10(4):e1003521 on April 3rd, 2014. PMID: 24699246. PMCID: PMC3974643. Link to publication page.
Core Facility: Computational Modeling Core
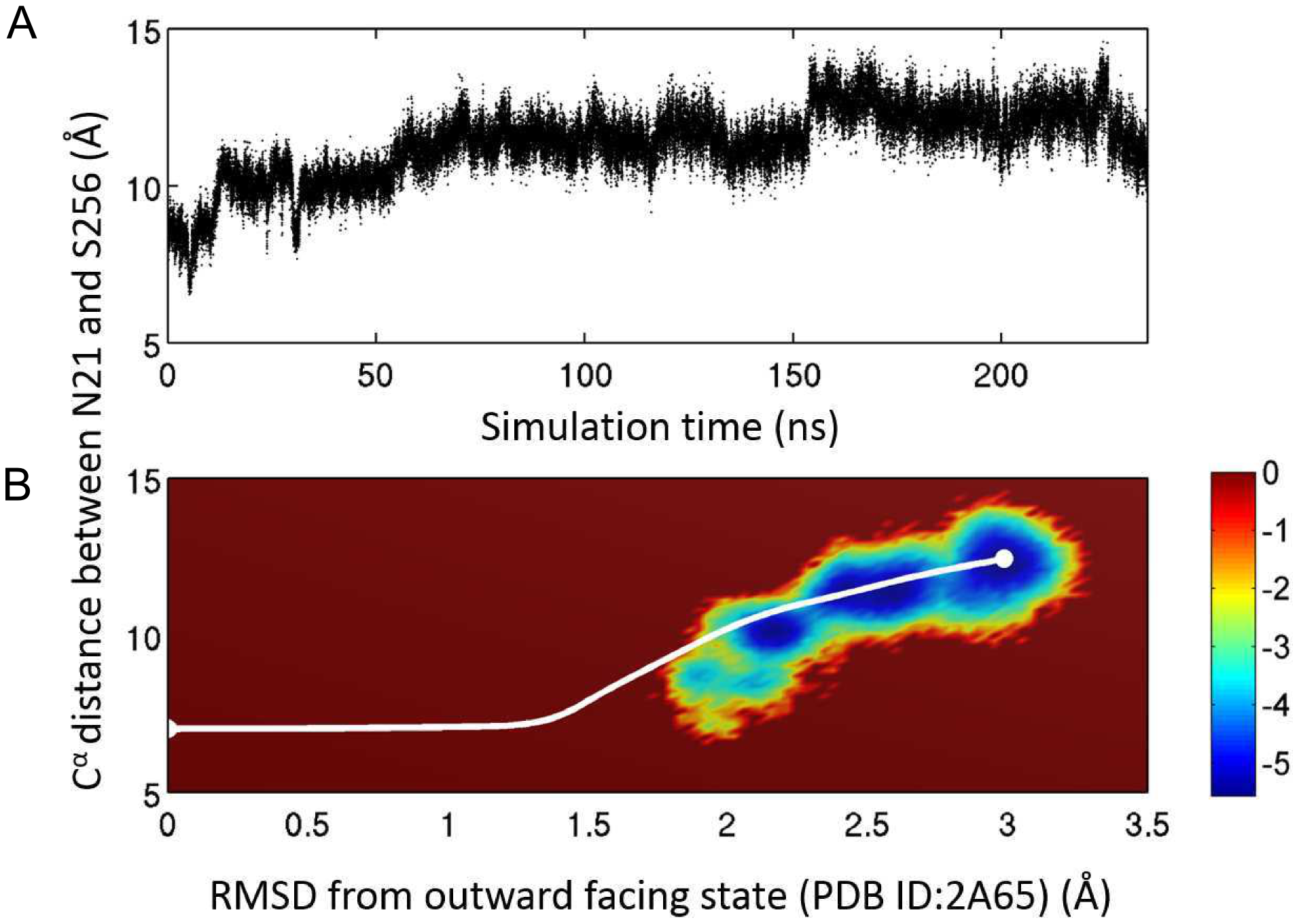
Figure 4. Comparison with all-atom simulation results. A. Time trace of the distance between atoms of residues 21 and 256 from a 235 ns long conventional MD simulation of the fully solvated system. The simulation was started from a structure obtained from a targeted MD simulation originated from the OFc state. The system undergoes a spontaneous transition to the IFo state. B. Comparison of the ANMPathway method and all-atom MD in the space of two order parameters. The all-atom MD results are shown as a pseudo free energy landscape
, where P is the 2D distribution. The color-scale goes from blue (low energy) to red (high energy). The pathway predicted by ANMPathway (white line) goes mostly through the low energy regions of the free energy landscape.
Abstract
Biomolecular conformational transitions are essential to biological functions. Most experimental methods report on the long-lived functional states of biomolecules, but information about the transition pathways between these stable states is generally scarce. Such transitions involve short-lived conformational states that are difficult to detect experimentally. For this reason, computational methods are needed to produce plausible hypothetical transition pathways that can then be probed experimentally. Here we propose a simple and computationally efficient method, called ANMPathway, for constructing a physically reasonable pathway between two endpoints of a conformational transition. We adopt a coarse-grained representation of the protein and construct a two-state potential by combining two elastic network models (ENMs) representative of the experimental structures resolved for the endpoints. The two-state potential has a cusp hypersurface in the configuration space where the energies from both the ENMs are equal. We first search for the minimum energy structure on the cusp hypersurface and then treat it as the transition state. The continuous pathway is subsequently constructed by following the steepest descent energy minimization trajectories starting from the transition state on each side of the cusp hypersurface. Application to several systems of broad biological interest such as adenylate kinase, ATP-driven calcium pump SERCA, leucine transporter and glutamate transporter shows that ANMPathway yields results in good agreement with those from other similar methods and with data obtained from all-atom molecular dynamics simulations, in support of the utility of this simple and efficient approach. Notably the method provides experimentally testable predictions, including the formation of non-native contacts during the transition which we were able to detect in two of the systems we studied. An open-access web server has been created to deliver ANMPathway results.

![Figure 2. Transition pathways displayed as a function of LID-CORE and NMP-CORE angles. Closed (bottom, square) and Open (top, diamond) substates of AK for the crystal structures (black) and their MD equilibrated (magenta) conformers. ANMPathway and three coMD [64] pathways are depicted as black, blue, magenta and green dots, respectively, with the arrows indicating the direction of reconfiguration in each case. The relative positions of the domains are defined similar to Beckstein et al. [72] and Gur et al. [64]. The NMP-CORE angle is the angle between the centers of mass of three segments I90-G100, L115-V125 and L35-A55, based on atoms and the LID-CORE angle is the angle between centers of mass of atoms of L115-V125, I179-E185 and V125-L153.](/site-media/images/publications/2014/das-2014-figure2.png)